Affinity Resin Selector
Find the perfect Ecolab
resin for your monoclonal
antibody purification needs.

Find the perfect Ecolab
resin for your monoclonal
antibody purification needs.

As a global leader in resin technology, we develop and manufacture small beads that are used in the most regulated industries in the world to separate, remove or recover very specific elements and compounds.
Learn More
With 40 years of manufacturing expertise and 30 years of regulatory experience, we supply leading separation, purification and extraction technologies to support chromatography applications within the Pharma and Medical space.
Learn more
We are a world leader in resin-based separation, purification and extraction technology, that provides sustainable solutions for our environment, businesses and healthcare.
Learn More
Ion Exchange Resins in Corn Sweetener Refining
Our always-ready technical support and service teams go the extra step to be your most trusted resource. We're here for you.


Demineralization of glucose syrup removes ash, protein and color from the solution. It also increases the long-term color stability of the syrup without the need for the addition of sulfur dioxide, which can cause a human allergic reaction in the final consumer products. Ash (calcium in particular) present in the 95% dextrose solution will have a negative effect on the performance of isomerase enzymes and must be removed by demineralization prior to isomerization of a typical 95% dextrose to a 42% fructose solution (42 High Fructose Corn Syrup (HFSC)).
Salts are added back into the 95% dextrose solution to facilitate isomerase enzyme performance and are removed after isomerization via demineralization to produce a pure sweetener for sale or for subsequent enrichment to a 55% fructose (55 HFCS) solution.
The ash content of glucose syrups is typically 0.25-0.45% by weight of total syrup dry solids and predominantly contains the following ions:
Sodium Na+, Calcium Ca++, Magnesium Mg++, Chloride Cl⁻, Sulfate SO4⁻ ⁻
To facilitate isomerization of dextrose to a 42% fructose solution, salts are added back to the syrup after demineralization and will be present in the 42% fructose. These salts must be removed prior to final evaporation or chromatographic separation. The ash content of 42 HFCS is typically 0.15-0.25% by weight of total syrup dry solids and consists primarily of:
Sodium Na+, Magnesium Mg++, Sulfate SO4⁻ ⁻, Sulfite SO3⁻
As the dextrose or fructose syrup solution passes through the resin bed, the sugars, ash, color bodies and proteins diffuse into the resin beads and can be exchanged or adsorbed onto the resin. In the strong acid cation bed, sodium, calcium, magnesium and other cations will replace the hydrogen ions on the resin due to their greater affinity for the resin than hydrogen ion. The syrup then passes through a bed of weak base anion resin where the mineral acids, organic acids and color bodies diffuse into the resin beads and are adsorbed onto the tertiary amine functional groups.

The weak base anion resin has a negligible mobile OH– counter ion which can be exchanged by an anion in solution. Without this ability to split neutral salts, the weak base anion resin must rely on the cation resin to produce acids for it to remove ions by acid adsorption. While the affinity of the cation resin for highly dissociated salts is good, the affinity for proteinaceous materials is much weaker and the cation resin will leak higher percentages of proteins during service.
To prevent proteins and organic acids from affecting the finished product quality, the syrup is typically passed through a secondary cation/anion pair for more complete removal of the impurities leaking from the primary demineralizer pair. Upon reaching the service exhaustion point of the primary demineralizers, the primary pair is regenerated, the secondary pair is moved into primary demineralization service and a third pair of freshly regenerated demineralizers is placed into secondary demineralization service.
Another equipment arrangement for glucose and fructose demineralization is a demineralizer two-bed pair coupled with a polishing strong base anion (Type II) mixed bed for even greater removal of the soluble impurities at the expense of some chemical efficiency. The higher product quality can increase isomerase enzyme life and provide a better feed for the fructose enrichment system. Upon exhaustion, the syrup is displaced from the resin bed and the resin is restored to a working condition with a chemical regeneration treatment. The metal ions are displaced from the cation resin by the passage of a strong mineral acid in an amount in excess of the stoichiometric exchange capacity of the resin in order to drive the equilibrium reaction. Hydrochloric acid is widely preferred to regenerate cation resins by displacing the metal ions and stripping off the proteinaceous compounds. The acids adsorbed onto the weak base anion resin are neutralized utilizing a base such as sodium hydroxide, sodium carbonate or aqueous ammonia. This acid neutralization is accomplished with a smaller excess of regenerant chemical than is required for the cation resin.
Regeneration of the resins is accomplished in a simultaneous sequence in a typical downflow service non-packed bed system as follows:
1. Syrup is displaced from the cation/anion pair by the introduction of condensate or demineralized water into the cation column which then pushes the syrup out through the anion. This step continues until the anion effluent syrup concentration has decreased to 0.1-0.5% dry solids.
2. Process water is passed in an upflow direction simultaneously in both the cation and anion columns to fluidize the resin.
3. A dilute hydrochloric acid solution passes through the bed exchanging hydrogen ions for the metal cations fixed onto the resin.
4. A dilute basic solution passes through the resin bed neutralizing and releasing the adsorbed acids.
5. At intervals of once per 5 to 25 cycles, strip off proteins adsorbed onto the cation resin utilizing caustic or ammonia. Organic acids adsorbed onto the anion resin are stripped utilizing an alkaline brine or hydrochloric acid solution.
6. Water passes through the resins and pushes out the regenerant chemical at a rate that ensures enough contact time for regeneration completion.
7. Condensate or demineralized water is used to rinse out the residual chemical in the beds.
8. Condensate or demineralized water passes through the cation and anion beds in series in a once-though or recirculating manner until the effluent quality reaches the desired conductivity limit of 10-30 microsiemens/cm (µS/cm).
9. Syrup is passed in series through the cation and anion beds displacing the rinse water. This ends when the anion effluent dry solids concentration reaches in excess of 95% of the feed syrup solids concentration.
| Demineralizer Sequence | |||
|---|---|---|---|
| Operation | Solution | Flow Rate (Bed Vols/Hr) | Volume (Bed Vols) |
| 1. Sweeten Off | H2O | 2.0-4.0 | 1.5-3.0 |
| 2a. Backwash Cation | H2O | 1.0 ft/min | 1.5-2.0 |
| 2b. Backwash Anion | H2O | 0.33 ft/min | 1.0 |
| 3a. Chemical in Cation | 7% HCl | 1.0-1.5 | 1.0 |
| 3b. Chemical in Anion | 4% NaOH | 1.0-2.0 | 1.5 |
| 4a. Cleanup Cation | 4% NaOH | 1.0-2.0 | 1.0 |
| (Alternate) | 3% NH3 | 1.0-1.5 | 1.5 |
| 4b. Cleanup Anion | 7% HCl | 1.0-1.5 | 1.0 |
| (Alternate) | 1% NaOH/10% NaCl | 1.0 | 1.5 |
| 5a. Slow Rinse Cation | H2O | 1.0-1.5 | 1.5 |
| 5b. Slow Rinse Anion | H2O | 1.0-2.0 | 1.5 |
| 6a. Fast Rinse Cation | H2O | 10-20 | 2.0-4.0 |
| 6b. Fast Rinse Anion | H2O | 10-20 | 3.0-5.0 |
| 7. Series Rinse | H2O | 5-10 | 2.0-5.0 |
| 8. Sweeten On | 30-50% DS Syrup | 2.0-4.0 | 1.0 |
| 9. Secondary Service | 30-50% DS Syrup | 2.0-4.0 | 30-60 |
| 10. Primary Service | 30-50% DS Syrup | 2.0-4.0 | 30-60 |
The demineralization equipment train typically consists of three cation/anion pairs or two cation/anion/mixed bed triplexes. The equipment utilizes food-grade rubber-lined carbon steel pressure vessels containing two or three sets of distributors made of CPVC or stainless steel and wrapped with polypropylene or a stainless-steel screen or Johnson wellscreen. In anion columns, which do not see a hydrochloric acid cleanup solution, stainless-steel distributors are often employed for greater strength against the large shrink/swell and pressure drop forces in the bed. Piping manifolds utilize polypropylene-lined carbon steel, rubber-lined carbon steel or stainless-steel pipe. Control of fluid direction is accomplished utilizing lined and stainless-steel plug valves, butterfly valves or diaphragm valves. Service flow can proceed either down through the resin beds, maintaining them packed in the lower half of the vessels, or up through the resins, maintaining them in a packed state in the upper half of the vessels.
Compartmentalized vessels with external backwash are also utilized. The vessels typically operate with a “water dome” (water fills the freeboard space between the middle and top distributors) to improve rubber lining life and minimize microbiological activity in the vessels during service.
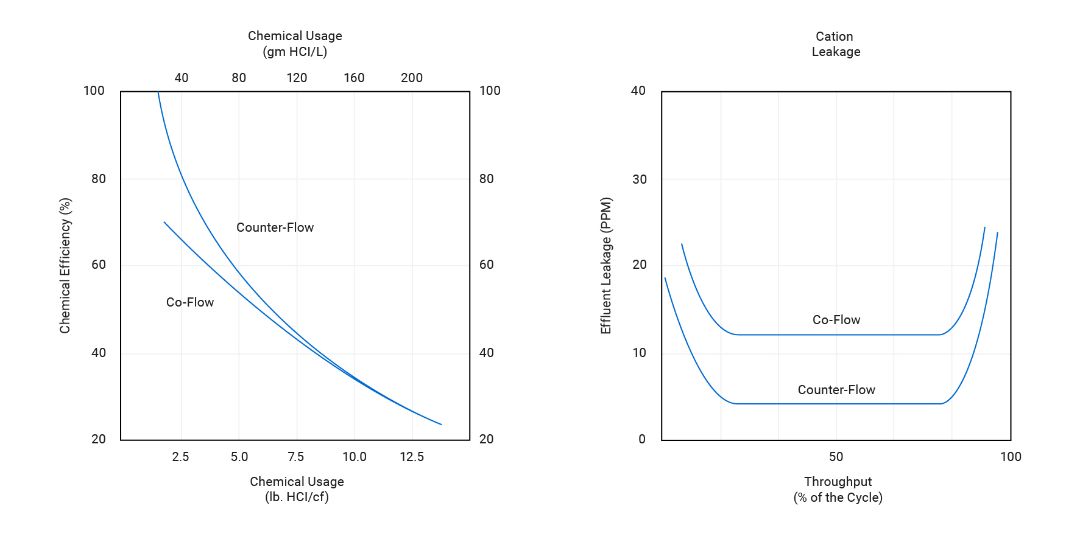
Improved product quality can be obtained from the cation resin by utilizing regenerant chemical passed in a direction counter to the service flow.
Since the syrup leaving the cation column is in equilibrium with the resin located at the outlet collector, the counter-flow passage of regenerant chemical creates an area of highly regenerated resin at the column effluent. At constant chemical dosage, the cation effluent quality is constant and contains fewer impurities than a co-flow regenerated cation resin bed.
Cocurrent and Countercurrent Cation HCl Efficiency and Leakage During Service Cycle
If the backwash step can be utilized infrequently, then the resin bed will be undisturbed during regeneration and a counter-flow regeneration may produce a high-quality syrup with the same throughput capacity at a somewhat lower chemical dosage. Since a weak base anion regeneration is an acid neutralization step and only a small excess of regenerant is required, no advantage is gained from counter-flow regeneration of the weak base resin.
Demineralization - Sweetener solutions
Purolite.com uses cookies to give you the best possible experience. By using Purolite.com, you consent to our use of cookies. If you do not wish to receive our cookies, adjust your browser settings. Read our Cookies Policy to learn more.