Affinity Resin Selector
Find the perfect Ecolab
resin for your monoclonal
antibody purification needs.

Find the perfect Ecolab
resin for your monoclonal
antibody purification needs.

As a global leader in resin technology, we develop and manufacture small beads that are used in the most regulated industries in the world to separate, remove or recover very specific elements and compounds.
Learn More
With 40 years of manufacturing expertise and 30 years of regulatory experience, we supply leading separation, purification and extraction technologies to support chromatography applications within the Pharma and Medical space.
Learn more
We are a world leader in resin-based separation, purification and extraction technology, that provides sustainable solutions for our environment, businesses and healthcare.
Learn More
Our always-ready technical support and service teams go the extra step to be your most trusted resource. We're here for you.
Colorants
Sugar colorants are a very complex mixture of organic compounds stemming from various sources. They can be categorized into two main types; natural colorants resulting from the cane plant and those developed during juice processing. The main characteristics of the colorant to be removed are that they are mainly hydrophobic (non-polar) and cover a large range of molecular weights. This results in exhibiting anionic behavior.
Natural Colorants
Flavanoids: < l kDa*
Melanins: >150 kDa
Chlorophylls, Xanthrophylls, Carotene: < l kDa
Colorants Developed During Juice Processing
Melanoldins, Maillard Reaction Products: < 50 kDa
Caramels: 25 kDa
HADPs: 1-5 kDa
* kDa is kiloDalton or a thousand Daltons. A Dalton is the atomic mass unit.

Decolorization with Ion Exchange Resin
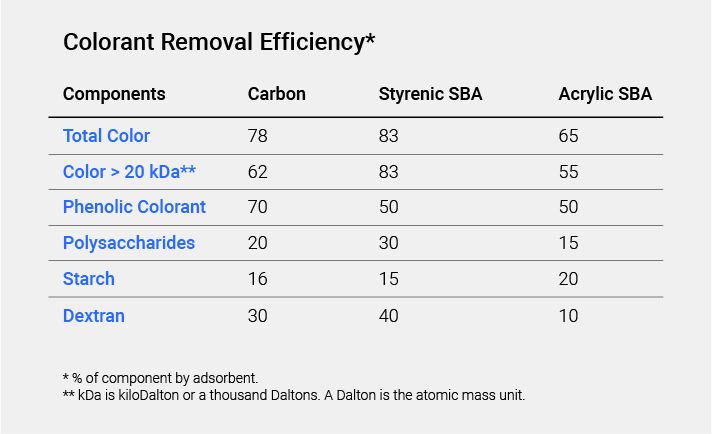
The resin matrix is usually composed of one of two types of polymeric material: polystyrenic with divinylbenzene crosslinks, resulting in a hydrophobic matrix, or a polyacrylic with divinylbenzene crosslinks which results in a more hydrophilic matrix.
Some refineries, for major decolorization, use only one resin type: acrylic or styrenic. Others employ an acrylic resin followed by a styrenic resin for polishing. The acrylic resin removes large molecular weight colored compounds which tend to foul the styrenic resin, providing good protection to the polishing resin.
Acrylic Resins
Acrylic resins have a slightly lower selectivity for sugar colorants but, unlike polystyrenic resins, they can be completely regenerated with sodium chloride solutions. This can be explained by the more hydrophilic character of the acrylic resin matrix. Even dark regenerant effluents from styrenic resins can be used to regenerate acrylic resins. The use of acrylic resin is justified when the color load is high and a low level of decolorization may be expected. Also, the use of a two-pass system gives a more constant level of decolorization than when only one resin bed is used.
Styrenic Resins
Styrenic resins are more prone to fouling by organic compounds than acrylic resins and need occasional acid regeneration. Also, their decolorization capacity is higher than that of acrylic resins, but the color is not so efficiently removed during regeneration and capacity can drop rapidly if overloaded.
Styrenic resins have a higher decolorization power because of their ability to fix colorants both through ionic bonds to the ionic active groups and through hydrophobic interactions with the resin matrix. Hydrophobic interactions increase with salt concentration, explaining the difficulty of using salt solutions to remove colorants fixed to the resin matrix.
Polystyrenic resins with an aromatic matrix should have a higher affinity for unsaturated colorants than acrylic resins. Styrenic resin types are favored when the inlet color is on the low end (i.e., up to 800 ICUMSA) and acrylic types when it is higher. Numerous plants today operate with a combination of both types, which is advisable when the inlet feed color fluctuates during the season.
Purolite.com uses cookies to give you the best possible experience. By using Purolite.com, you consent to our use of cookies. If you do not wish to receive our cookies, adjust your browser settings. Read our Cookies Policy to learn more.