Affinity Resin Selector
Find the perfect Ecolab
resin for your monoclonal
antibody purification needs.

Find the perfect Ecolab
resin for your monoclonal
antibody purification needs.

As a global leader in resin technology, we develop and manufacture small beads that are used in the most regulated industries in the world to separate, remove or recover very specific elements and compounds.
Learn More
With 40 years of manufacturing expertise and 30 years of regulatory experience, we supply leading separation, purification and extraction technologies to support chromatography applications within the Pharma and Medical space.
Learn more
We are a world leader in resin-based separation, purification and extraction technology, that provides sustainable solutions for our environment, businesses and healthcare.
Learn More
항상 준비된 기술 지원 및 서비스 팀은 가장 신뢰할 수 있는 리소스가 되기 위해 한 단계 더 나아갑니다. 우리는 당신을 위해 여기 있습니다.
Originally presented at Society for Mining, Metallurgy & Exploration, February 19-22, 2012.
Mikhail Mikahaylenko
Purolite, Moscow, Russia
A. Blokhin
St.-Petersburg State of Inst. of Tech., St.-Petersburg, Russia
Editor's Note:
* Since the publication of this paper, several product names have been rebranded and their names have changed versus what was initially noted in this paper. Product names are currently:
Purolite® A110 (formerly Purolite D5248)
Purolite A500Plus (formerly Purolite A500/3612)
PurogoldTM MTA1930 (formerly Purolite A100Mo)
PurometTM MTA1701 (formerly Purolite A170/4675)
Puromet MTA1721 (formerly Purolite A172/4635)
Puromet MTS9500 (formerly Purolite S950)
Puromet MTS9570 (formerly Purolite S957)
Puromet MTS9840 (formerly Purolite S984)
Introduction
Rhenium, molybdenum and tungsten are the elements demanded by high-end technologies.
These metals are found in process solutions in forms of their oxyanions: ReO4-, MoO42- and WO42-. The latter two can exist in forms of iso- and/or heteropolyanions. Basically, all three metals demonstrate very similar chemical behavior in aqueous solutions. Unfortunately, in their mineral sources, these elements commonly accompany each other, like pairs of rhenium-molybdenum or molybdenum-tungsten. Their economical separation is a challenging task.
The main problem associated with rhenium recovery is its low concentration and interference with other ionic constitutes in process solutions, especially with molybdenum.
It is known that molybdate and tungstate tend to form large polymeric anions. Their size, or polymerization rate, depends on pH and concentration of the solution. Pope [1] summarized research information on the state of molybdenum and tungsten at different pH.
At pH above 7-7.5 only monomeric molybdate anion MoO42- exists.
In pH range from about 6 down to 2.0 there are polymerized forms, such as Mo8O264-.
At pH below 2.0 cationic forms MoO22+, Mo2O52+ and Mo3O82+ appear. The anionic and cationic forms of molybdenum coexist in dynamic equilibrium. The share of molybdenum, which is in cationic form, increases with increasing of the solution acidity. At the same time acid complexes like MoO2(SO4)22– appear as well.
Ionic forms of tungstate can be the following.
At pH >8, a dominating form is normal tungstate WO42-. In pH range from 6 to 4, heptatungstate W7O246- and dodecatungstate H2W12O4210- present in the solution. Protonated forms like H3W12O429- and H4W12O428- also present the later.
At pH <4, the following anionic forms exist: H2W12O406-, H3W12O405-.
Stronger acidification leads to further protonation and formation of neutral molecules like H12W12O42. Such larger neutral molecules are poorly soluble and this is why, at pH below 2, tungsten almost entirely precipitates as tungstic acid.
Increasing of concentration facilitates the polymerization trend. At concentration below 10-6 M only monomeric forms like WO42-, HWO4- and H2WO4 exist.
Molybdate and tungstate can also form mutually mixed polyanions.
Rhenium (VII) in aqueous solutions has a stable form of monomeric anion ReO4- in full range of pH.
Different processes have been developed and used in industry for such specific tasks as selective rhenium recovery from different process solutions, separation of rhenium from molybdenum, removal of molybdenum impurities from rhenium and tungsten from molybdenum, recovery of molybdenum and tungsten from their concentrates, conversion of sodium salts of tungstic acids to its ammonium salt as well as the processing of recycled raw materials. Not all of them are economical because of numerous process operations, large consumption of specific reagents etc.
The presented paper reviews application of the ion exchange resins, shortly described in the table below, for separation and purification of rhenium and tungsten.
Recovery of Rhenium
There are few general sources of rhenium. Major part of rhenium production comes from processing of mineral resources. Commonly this metal is recovered as by-product at smelting of copper concentrates or rousting of molybdenite concentrates. Volatile rhenium heptoxide Re2O7 is trapped by off-gas cleaning systems and appears in the sulfuric acid solutions in form of perrhenate anion.
Molybdenite calcine also contains some residual rhenium and the later can be leached out by sulfuric acid.
Thus, depending on origin of the rhenium pregnant solution, it can contain different portions of molybdenum as well.
Other significant source of rhenium is secondary materials coming from spent Pt-Re catalysts and scrap of Ni-Re super alloys. Such materials are usually processed with use of hydrochloric, sulfuric, nitric acid or their different combinations.
In aqueous solutions Re (VII) exists in form of very stable perrhenate anion ReO4-. Like other large monovalent anions the perrhenate has strong affinity to anion exchange resins. Perrhenate sorbed by strong base anion (SBA) exchange resins can be eluted under harsh conditions with use of large volumes of 6N nitric acid, 1N perchloric acid [2, 3], aqueous-organic solvents systems [3], metal chlorides in acids [4], thiocyanate in acid [8].
In contrary to case of SBA resin, rhenium sorbed by weak base anion (WBA) exchange resins is readily eluted by alkaline solutions.
Other technical challenge in rhenium recovery is large excess of competing anions like sulfates, nitrates, chlorides and others. So, a must for the WBA resin is high rhenium selectivity.
The table below compiles several examples of typical primary rhenium solutions. It is seen that concentrations of rhenium and its ratio to molybdenum concentration can vary widely. This is one of the decisive parameters for choice of proper resin.

Separation of rhenium from molybdenum in primary solutions
Use of SBA resins for collective sorption of rhenium and molybdenum followed by selective desorption of molybdenum by alkali and rhenium by concentrated nitric acid has been suggested earlier [5]. We would like to introduce a resin which is able to selectively separate rhenium from molybdenum in the solution and which resin is easily eluted by ammonia solution.
For our consideration, it is important that under low to strong acidic conditions molybdenum exists as very large anions or in cationic form while rhenium is presented by much smaller ReO4- anion. By use of sieving effect, the latter can be effectively separated from molybdenum by gel type resin Puromet MTA1721.[6, 7]
Due to smaller size of anion, rhenium easier diffuses inside of the resin bead. Large molybdenum anions sorbed mostly by surface of the resin. Puromet MTA1721 resin and artificial solutions were used in the kinetics study.[6]
Investigation of kinetics was undertaken in static mode. The study was run in seven beakers per experimental point. To simulate virtually constant concentration of metals in the outer solution such large volume of the later to the resin as 2,000 to 1 was used. Composition of the artificial solution was the following, mg/l:
Mo – 3200;
Re – 12.5;
P – 70.0;
Cl – 16.0;
SO42- – 100 g/l;
pH = 1.2.
Phosphate anions were to simulate arsenates.
As the kinetic curve on the figure below shows, molybdenum reaches maximal loading in about 4 hours of contact while rhenium continues loading up to about 25 hours.
Then sorption study in column was performed on the similar solution, mg/l:
Mo – 3426;
Re – 18.9;
P – 70.0;
Cl – 16.0;
SO42- – 100 g/l;
pH = 1.5.
Flow rate of the feed was 10 bed volumes per hour (BV/hour).
Kinetics Study for Sorption of Rhenium and Molybdenum by MTA1721 Resin

As one can see, saturation of the resin in dynamic experiment with molybdenum occurs very soon while rhenium loading is much longer. Thus the resin is separating rhenium from the molybdenum rich feed.
Metal loadings on the resin were 10.45 g/l for rhenium and 1.62 g/l for molybdenum.
Form of the breakthrough curve evidences that rhenium sorption process has very slow kinetics. It can be explained by two reasons. Firstly, large perrhenate anion slowly diffuses in inner volume of the gel polymer matrix. Secondly, multi-charged molybdate anions adsorbed on the surface of the resin beads can form a kind of shield preventing penetration of other negatively charged ions into the bead.
The saturated with rhenium and molybdenum resin was again packed in column for desorption experiment. Desorption solution was fed upflow. Concentration of NH4OH in eluent was 50 g/l. Desorption flow rate 30 ml/hour (about 1 BV/hour). Process temperature 60 °C. Outlet solution was periodically analyzed for the metals concentrations.
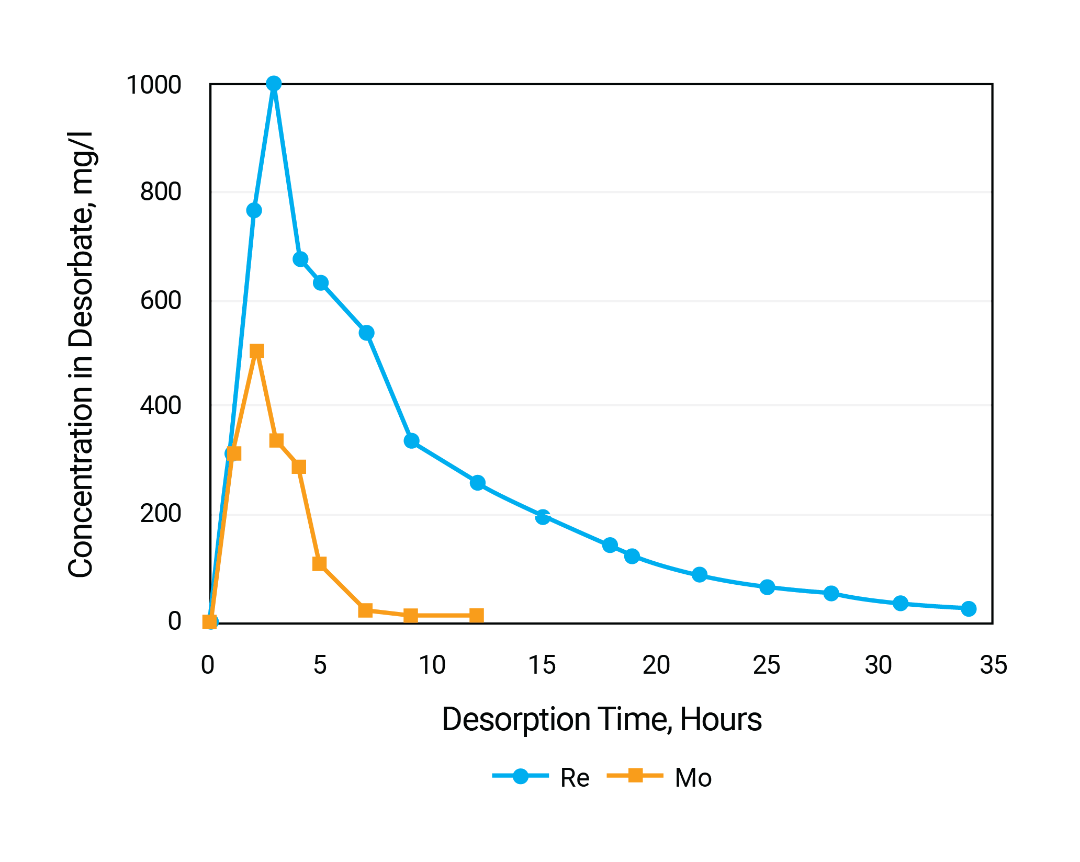
Desorption of rhenium from the gel resin is slow what can be also explained by slow diffusion of perrhenate anion from inner part of the polymer beads to outside.
Residual capacity of the resin for rhenium is 0.83 g/l and for molybdenum just 0.07 g/l.
Concentrations of metals in entire desorbate were 15.4 mg/l for molybdenum and 766 mg/l for rhenium. Thus, separation number S for rhenium over molybdenum from the feed to desorbate was as high as:

Assuming that molybdenum is mostly adsorbed on the surface of the resin the separation number can be further improved via two-step desorption. At first step, the most of molybdenum is to be flushed out by fast and short rinsing by diluted alkali. Then, at the second step, rhenium is to be desorbed by the slower flow of more concentrated alkali.
Sorption of rhenium from different acids in absence of molybdenum
Nowadays significant amount of rhenium comes from copper smelters. Their off-gas treatment systems generate rinsing and produced sulfuric acid streams containing rhenium. These streams do not contain a meaningful concentration of molybdenum. To date, there are three copper smelter plants in the world, which have installed Puromet MTA1701 resin for rhenium recovery from sulfuric acid.
The figure below demonstrates the simplest example of rhenium sorption from sulfuric acid by macroporous WBA resin Puromet MTA1701. Affinity of the Puromet MTA1701 resin for rhenium remains high at different concentrations of sulfuric acid.
For other experiment, the solution of the following composition was used: Re – 0.85 g/l; Na2SO4 – 103 g/l; pH 3.1. Full capacity for rhenium on Puromet MTA1701 resin, achieved in column experiment, was 162.2 g/l. Desorption from the loaded resin was done with 12.5% ammonia solution. Full rhenium desorption could be done with less than 10 BV of the eluent. Rhenium concentration at the pick of elution profile reached 35 g/l. Rate of desorption was above 94%.
When such rhenium sources are processed as spent catalysts or superalloy scraps, there is no such problematic impurity as molybdenum. Common metals in the process solutions are nickel, aluminum, iron etc. They do not interfere with sorption of rhenium, which is in anionic form. However such solutions have different anionic background. Often nitric or hydrochloric acids are used, either as individual leaching agents or as their mixture. So, it is important to know how rhenium is recovered from these acids.

Sorption of rhenium from solution with chloride matrix remains very effective. Isotherms in the figure below show similar capacities as they are obtained at sorption from sulfuric acid.

Application of SBA resins for rhenium recovery is known for long time.[2-5, 8] Purolite A600 product is a Type I, SBA resin on gel matrix with chloride exchange capacity around 1.5 eq/l. This resin was compared with A170* for rhenium sorption from nitric and hydrochloric acids of different strength at initial rhenium concentration 100 mg/l.

Rhenium sorption on Puromet MTA1701 resin is less sensitive to chloride presence than common SBA resin.
Nitrates depress sorption of rhenium much stronger than chlorides. As it is clear from the figure below, both resins demonstrate very similar capabilities for rhenium sorption though Puromet MTA1701 still has some advantage. But one should remember that desorption of rhenium from SBA resin is much harder [2-5, 8].

Recovery of Molybdenum
Sorption of molybdenum from concentrated acids
Some manufactures are to deal with nitric-sulfuric molybdenum solutions generated by etching processes. Acid concentration in obtained solutions is high. So, recovery of molybdenum from such solutions may have practical interest.

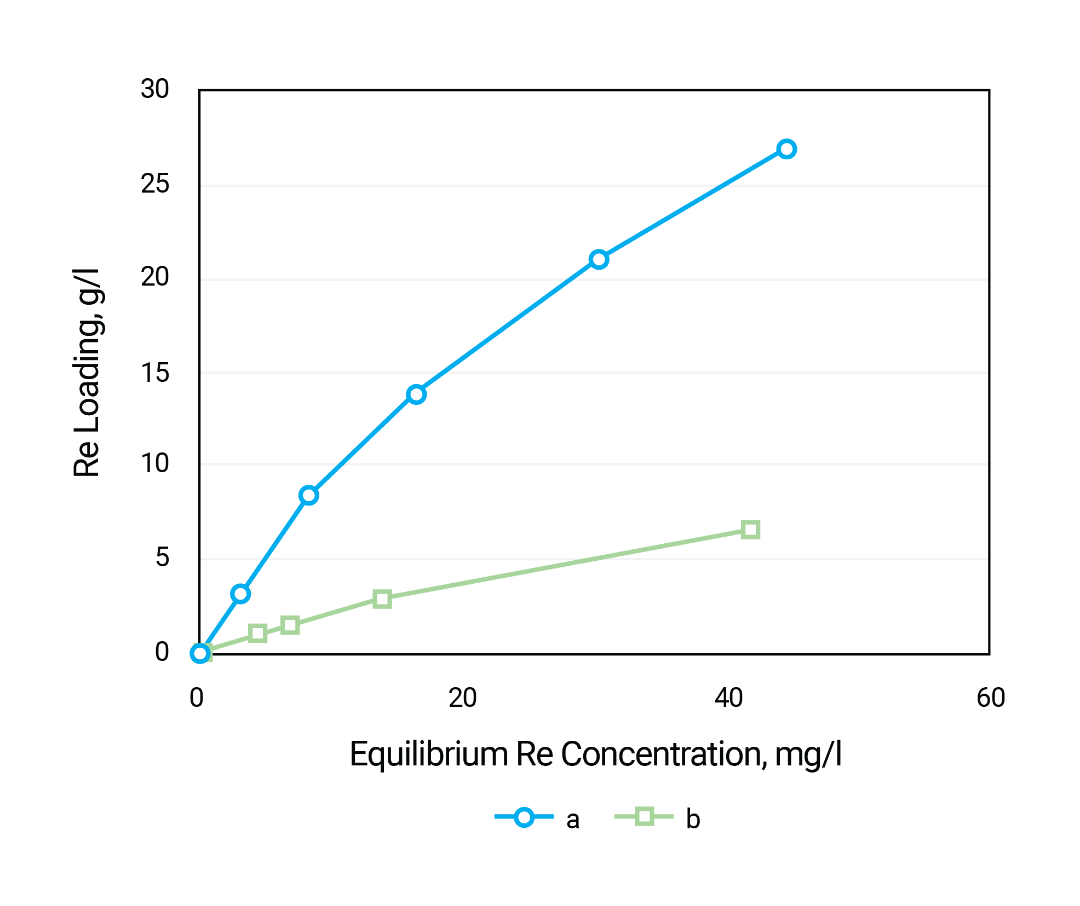
Three resins were compared for this task. The figure above represents isotherms of molybdenum sorption from mixture of 1M HNO3 and 2M H2SO4.
First of all, it is clear that anion exchange resin Purogold MTA1930 has much less sorption capacity under such acidity than two other products. It can be explained by two reasons. Firstly, high concentrations of bisulfate and sulfate anions, and especially nitrate anions, strongly compete with anionic forms of molybdenum for ion exchange sites. Secondly, under such high acidity molybdenum mostly exists in cationic forms, which are not sorbed by anion exchange resin.
Isotherm of sorption for Puromet MTS9500 resin has very convex upwards profile at lower concentrations of the metal. At the same time this resin becomes not sensible to increasing of molybdenum concentration above 2.5-3 g/l; the metal loading on the resin remains the same.
At some molybdenum concentration the capacity of Puromet MTS9570 resin becomes similar to Puromet MTS9500 and exceeds the later with further concentration increasing.
Thus Puromet MTS9500 resin can be recommended for molybdenum recovery from its lean solutions in concentrated acids while Puromet MTS9570 resin is more suitable at higher concentrations of the metal. Another advantage of Puromet MTS9500 resin can be better tolerance to presence of ferric iron under so high acidity. However, the later assumption is to be verified by experiment yet.
It is interesting to notice that Puromet MTS9500 resin has as phosphonic acid groups and weak base anion exchange function as well. Possibly both acid and base functions of the resin make input in its high molybdenum selectivity.
At sorption from 100 g/l sulfuric acid solution, the Puromet MTS9570 resin capacity for molybdenum reaches 80 g/l at equilibrium concentration of the metal 2 g/l.
Desorption can be effectively done by about 5 BV of 15-25% ammonia providing a rate of desorption above 90%. The second figure below demonstrates elution profiles for these two resins. They were loaded with molybdenum in columns from solution with molybdenum concentration 20 g/l. Full molybdenum capacity was 84 g/l for Puromet MTS9570 resin and 64 g/l for Puromet MTS9500.
Presumably, molybdenum sorption by resins with phosphonate functionality has cation exchange mechanism. However, the formation of heteropoly acid function via condensation of molybdate anion with phosphonate groups should not be disregarded as well.
Indirectly, the later statement can be supported by the fact of very effective desorption of molybdenum from this resin by aqueous ammonia. In this case, the heteropolyacid structure is to be hydrolyzed under alkaline conditions and produced molybdate anions are repulsed from the cation exchange, i.e., negatively charged matrix.
Regardless, the sorption mechanism of the Puromet MTS9570 and Puromet MTS9500 resins can be used for molybdenum recovery from concentrated nitric and sulfuric acids.
Elution of molybdenum from these resins can be easily done by ammonium solution (figure below).

Processing of molybdenite concentrates
Block scheme in the figure below represents a generalized flowsheet for recovery of molybdenum from concentrates via autoclave leaching.

Conditions of leaching can be varied by pressure, nitric acid concentration, temperature etc. If leaching is curried out under ambient pressure then a higher concentration of nitric acid is required.
The autoclave leaching of molybdenite concentrates results in increasing of total acidity due to the oxidation of sulfide minerals.
Purogold MTA1930 resin can be used in the process. Before the first sorption stage acidity of the leachate is corrected to pH 0.8-0.9. Sorption at so low pH is required to avoid co-precipitation of molybdenum with iron. At this stage, most of the molybdenum is extracted from the leachate. The sorption contact time for the resin is around 2.5-3 hours.
Molybdenum loading of the resin depends on the concentration of molybdenum in the feed and is affected by nitrate anion. The typical resin capacity for molybdenum is about 90 g/l at the first stage.
After further correction of pH to 2–2.3, the second sorption can be done. At this stage, resin capacity increases to approximately 120 g/l as per molybdenum.
Full desorption can be effectively done by about 5 BV of 15–25% ammonia. Desorbates can have molybdenum concentration up to 75 g/l and, after additional concentrating can be used for crystallization of ammonium paramolybdate (APM).
Due to proper bead size and good mechanical stability, the Purogold MTA1930 resin can be used for sorption from filtrated “clean” solutions or directly from pulps. Elimination of solids separation operations in the resin in pulp (RIP) process significantly reduces costs.
Conversion of Sodium Paratungstate (SPT) to Ammonium Paratungstate (APT)
In hydrometallurgy, the tungsten mineral concentrates can be processed via alkaline (soda ash) or acidic routes. At either route, the conversion of SPT to APT may be required for further production of pure tungsten compounds, items and alloys.
One of the methods to do this operation is the application of ion exchange resins.
Processing of carbonate PLS
Carbonate routes for the processing of tungsten mineral concentrates produce solutions with a high tenor of sodium carbonate.
So, an investigation of tungstate sorption from concentrated sodium carbonate solutions was undertaken. Strong base anion exchange resins must be used for such kinds of solutions.
Increasing carbonate concentration in the feed solution affects the loading capacity of the resin for tungsten.
The figure below illustrates the ability of Purolite A500Plus resin to uptake tungsten from carbonate solution. It can be noticed that there is no significant concentration of the metal on the resin. The effect of carbonate concentration on tungsten resin capacity is demonstrated by the second figure below.

Desorption of tungsten from Purolite A500Plus resin can be effectively done by mixture of 60-110 g/l of NH4HCO3 and 140-200 g/l of (NH4)2CO3. Desorption rate is above 95% (the table below).
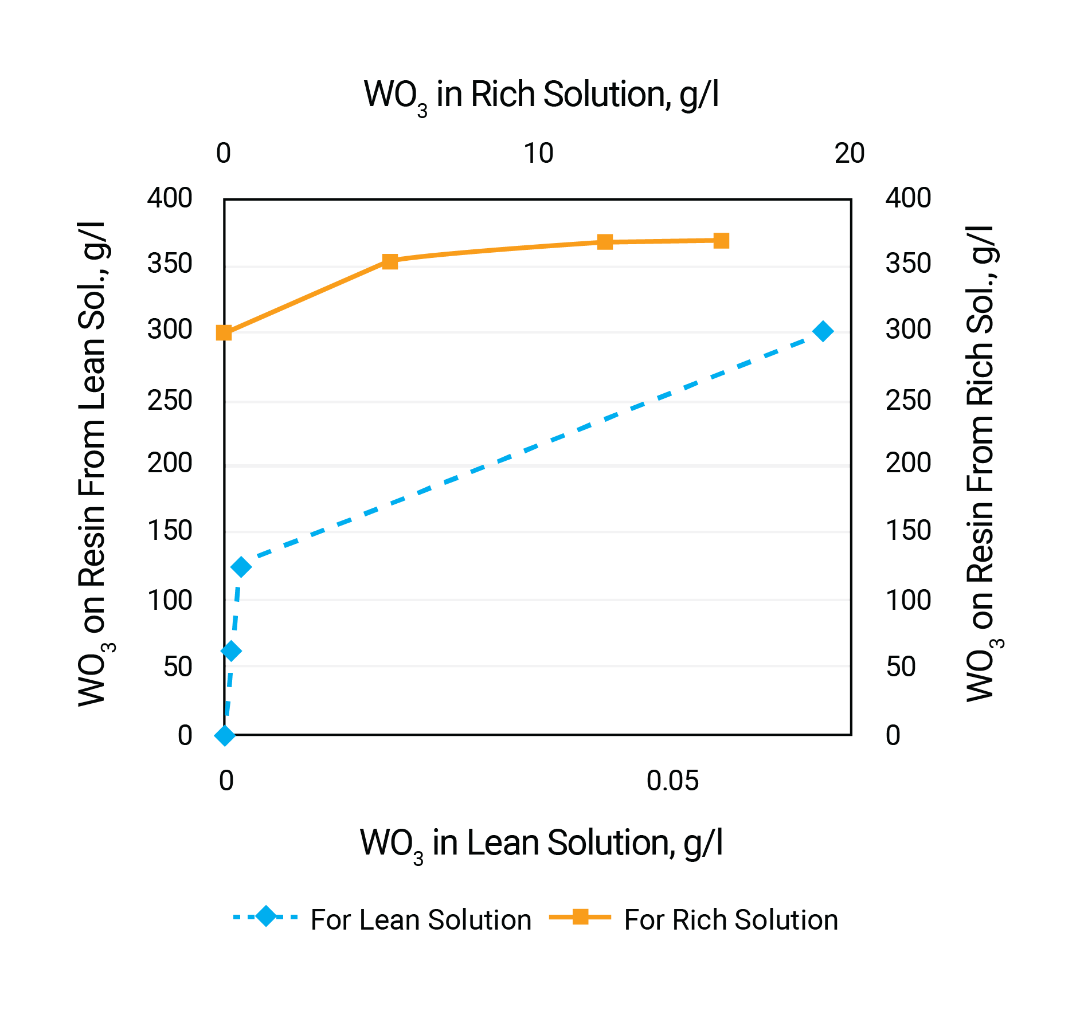
Obtained in desorbate ammonium salt of tungstic acid can be concentrated and separated via crystallization.
Processing of acidic PLS
Direct ion exchange sorption of tungsten from carbonate solutions is not so effective because of relatively low capacity of the resin under alkaline conditions.
Another route implies acidification of the pregnant solution. It leads to polymerization of tungstate anions. Sorption of polymerized anions significantly increases operating capacity of the resin.
Sorption from acidic solutions also implies application of weak base anion exchange resins. Thus elution can be easily done by aqueous ammonia yielding rich APT. The later is more suitable for further concentrating and crystallization of APT. Results of Purolite A110 resin testing for tungsten recovery from sufuric acid solution is presented in the figure below.
Sorption was curried out from solution of 0.5M Na2SO4 at pH 3 adjusted by sulfuric acid. It can be seen from the isotherms that investigated resin has high affinity for tungstate anions. Distribution coefficient at equilibrium concentration 0.07 g/l is as high as 4,500. As far as WO3 concentration increases above 12-15 g/l the resin capacity growth is declining and remains almost constant at level of about 375 g/l.
Desorption of tungsten from the Purolite A110 resin can be done by aqueous ammonia with rate more than 90%.
So, at processing of rich solutions this resin does a conversion work of SPT to APT without concentrating of tungsten in desorbate.
However, if this resin is used for processing of acidic lean solutions of tungsten then concentrating parameter for this metal can be as high as several hundred times.
Purification of Perrhenate and Paramolybdate Solutions
Removal of molybdenum impurity from rhenium
In practice different secondary materials can be produced at processing of molybdenum or copper ores, which materials can contain relatively high proportions of rhenium. Such materials can bear different impurities like base metals large proportions as well.
When pregnant solutions have relatively high ratio of rhenium to molybdenum then it can be interesting to remove molybdenum first for downstream recovery of rhenium.
The artificial solution has been used for investigation of possibility to remove molybdenum impurity from rhenium solution (table below).

Sorption test was run in a column packed with 200 ml of Puromet MTS9570 resin. Flow rate of the feed was 70 ml/hour. As it was assumed, the Puromet MTS9570 resin did not uptake rhenium from solution but loaded molybdenum and ferric iron. Copper sorption was negligible.
In this process a separation factor of molybdenum over rhenium fixed in the resin was the following:
Then desorption was done by 5% solution of ammonia at 1 BV per hour (see table below). Time of desorption was about 12 hours; yield of desorbate about 10 bed volumes (BV).

Of course, this approach to the separation of molybdenum from rhenium can not be directly applied to those real solutions, which contain ferric iron because Puromet MTS9570 resin binds the iron too strongly. However, if to undertake measures for reduction of iron to divalent state before sorption, then this resin can perform well. Alternatively, the sorbed ferric iron must be eluted by 6N hydrochloric acid either by another suitable reagent to recover the resin capacity.
Removal of tungsten impurity from molybdenum
Tungsten contamination affects the ductility of molybdenum. Molybdenum to be used for the production of wire, foil and other rolled materials must have less than 0.01% of tungsten. Production of molybdenum monocrystals requires less than 10-4-10-5% of tungsten impurity.
The problem is that both metals are able to form cross-contaminated polyanions. However, under definite acidity, tungsten tends to form isopolyanions, while molybdenum yet remains mainly in a monomeric form. This minor difference in the behavior of molybdenum and tungsten in aqueous solutions makes the use of ion exchange possible for the removal of tungsten from molybdenum.
After screening work done among many candidates the Puromet MTS9840 resin has been chosen as the most effective material for molybdenum purification.
Investigation of sorption isotherms (the figure below) demonstrated that Puromet MTS9840 resin has high potency for selective sorption of tungsten from the concentrated molybdate solution.

For sorption in column the following composition paramolybdate and paratungstate in water was used: Mo – 140 g/l; W – 0.9 g/l. The feed flow rate was 1 BV/hour. Main results of the refinery process are shown in the table below.

Under the given conditions, the breakthrough occurs after the 26th BV. It provides convenient operation and considerable tungsten loading on the resin.
A desorption test was done by ammonia solution for a resin sample loaded with tungsten in the column experiment. Desorption smoothly run with a small volume of desorbent (see figure below).
Molybdenum is sorbed together with tungsten by the resin. As per analysis of desorbate the resin capacity for molybdenum is 70 g/l. It corresponds to about 1.2% of total amount of molybdenum passed the column with the feed solution.
As a useful benefit to the process of tungsten removal, the Puromet MTS9840 resin also allows lessening the content of base metals impurities in the molybdate solution (see table below). The resin functionality has dual nature. In addition to anion exchange properties, it can work as a chelating resin as well. This is why this resin is able to uptake cations of transition metals underused conditions.
Conclusion
An important facet, common for all considered resins and processes, is the fact that all of them directly produce ammonium salts of rhenic, molybdic and tungstic acids. The ammonium salts are the most important commodity forms of these metals because they can be easily converted to pure metals and alloys.
Ion exchange provides for these metals the shortest way from the pregnant solutions to the ammonium salts. This is especially fair in case of separation of rhenium from molybdenum. Other tasks dealing with low concentrations of rheniumand other considered here metals could be economically solved by ion exchange sorption only.
Thus, it can be stipulated that there is a powerful arsenal provided by ion exchange resins for manufacturing of pure rhenium, molybdenum and tungsten compounds from raw sources of different origination.
References
Purolite.com uses cookies to give you the best possible experience. By using Purolite.com, you consent to our use of cookies. If you do not wish to receive our cookies, adjust your browser settings. Read our Cookies Policy to learn more.